A study by a TFRI-funded team based at the Vancouver Prostate Centre has found that silencing a key driver of castrate-resistant prostate cancer and regulating cell division (mitosis) can improve patient drug response.
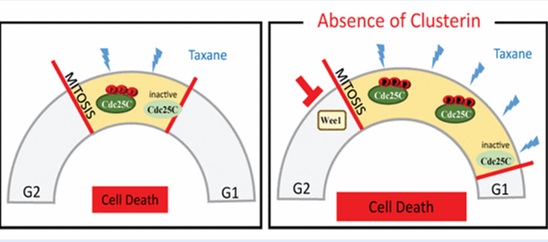
The present study, led by Dr. Martin Gleave and published in EMBO Molecular Medicine http://onlinelibrary.wiley.com/doi/10.15252/emmm.201506059/full (May 2016), explored the biological effects of clusterin (CLU) on mitosis, finding that silencing CLU-induced activation of Cdc25C makes cancer cells more sensitive to mitotic-targeting agents such as chemotherapy.
Therefore, drugs that can be used to inhibit CLU could improve patient’s treatment response when used in combination with standard treatments. CLU is a stress-activated molecular chaperone that can activate treatment resistance to taxanes (drugs that block cell division).
Further, the present study authored by Nader Al Nakouzi demonstrated for the first time that resistance to taxanes may occur via Cdc25C-Wee1-MPF regulation, and that co-targeting Wee1 induction caused by inhibiting CLU could be a new and effective method for overcoming drug resistance in prostate cancer patients and may also increase survival rates.
Prostate cancer is the third leading cause of cancer-related death in Canadian men, and one of the most common cancers for this demographic. It initially responds well to hormone therapy, but can often become resistant to treatment, an outcome that is linked to poor prognosis.
This study data complements prior work by Dr. Gleave’s team in developing anti-clusterin drugs to overcome treatment resistance and increase survival for patients. It also builds on other project work related to other drugs currently in clinical trials, and therapies that inhibit Hsp27 and ERG, which encode proteins typically mutated in cancer.
Study: Clusterin knockdown sensitizes prostate cancer cells to taxane by modulating mitosis
Authors: Nader Al Nakouzi, Chris Kedong Wang, Eliana Beraldi, Wolfgang Jager, Susan Ettinger, Ladan Fazli, Lucia Nappi, Jennifer Bishop, Fan Zhang, Anne Chauchereau, Yohann Loriot & Martin Gleave
Funding: This project was supported by a Terry Fox New Frontiers Program Project Grant TFF116129, a Prostate Cancer Foundation of British Columbia (PCFBC) Grant 2013, and a Prostate Cancer Canada Team Grant T2013-01. We would like to thank Mr. Dulguun Battsogt for his technical support and Mr. Robert Bell for his help with the statistical analysis.
Links #2