A novel technology has helped a TFRI-funded multidisciplinary research team identify how a key driver of human cancer works, cracking open the possibility for new targets and treatments to stop it in its tracks.
MYC mutations are present in over half of all cancers, and whenever they occur, cancers are more aggressive, and prognosis is much worse, explains Dr. Linda Penn, a TFRI-funded investigator and molecular oncologist at the Princess Margaret Cancer Centre in Toronto. “Whenever MYC activity is out of control, it's a very nasty beast. In fact, if MYC is overexpressed in a cell, it can drive cancer in that cell, no matter what type of cell it is.”
But despite three decades of research, scientists have been unsuccessful in identifying drugs that could inhibit it, partly because there was little understanding of how MYC actually worked.
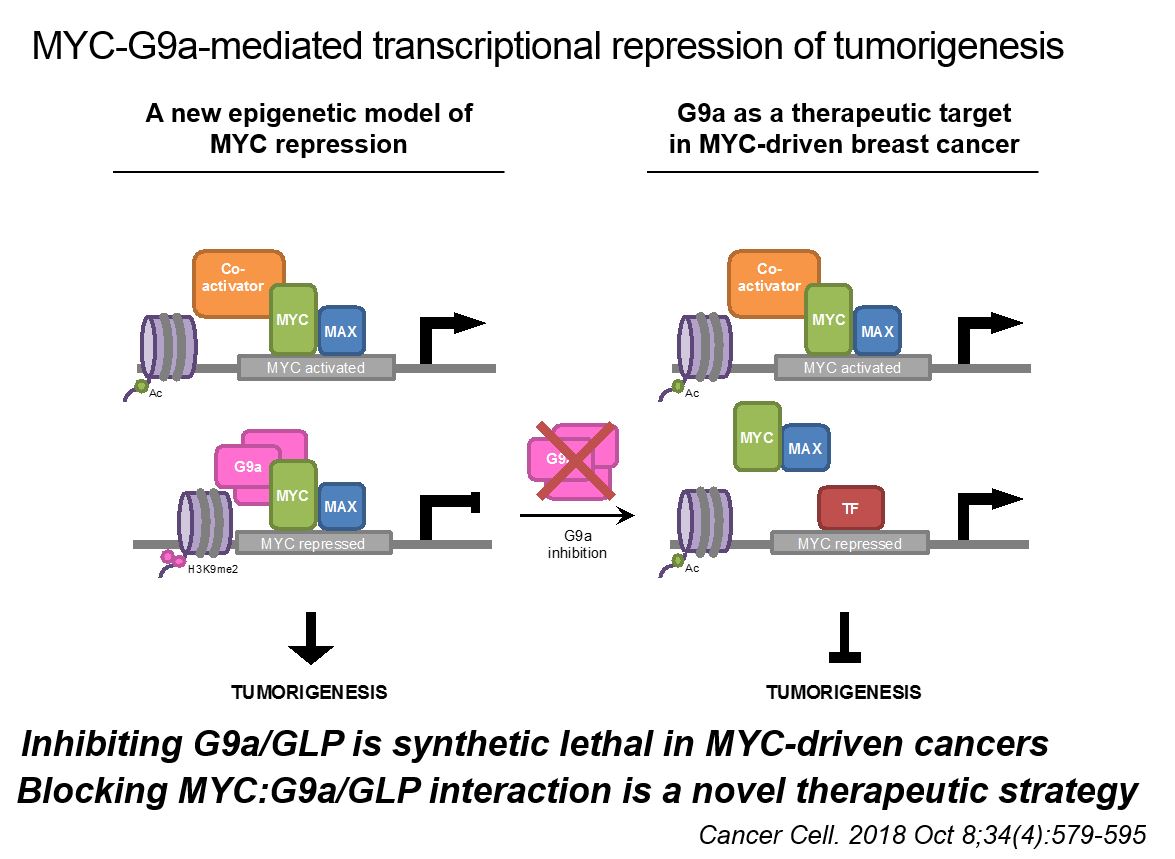
Now Dr. Penn and her multidisciplinary colleagues are closing this knowledge gap with a new study published in Cancer Cell (November 2018) that explains MYC’s interaction with a protein called G9a. “What we learned is that the interaction of MYC with G9a is extremely important for MYC to drive oncogenesis. This is an important finding because it means that if we figure out how to target G9a, we could basically disable MYC activity,” says Dr. Penn, the study’s senior author.
An epigenetic regulator, G9a helps determine what bits of the genome are turned off or turned on. The study team found that when it interacts with MYC, it represses specific gene transcription causing cancers to form.
To make this breakthrough, the team used a novel new technology known as BioID to identify all proteins that interact with MYC.
“This collaborative effort with this new technology has allowed us to just bust open the cell for the first time and identify all those partner proteins that make up the MYC interactome,” says Dr. Penn. “And G9a isn’t the only one we have identified – there are 350 other proteins that MYC interacts with that can be analyzed as potential targets for the development of anti-cancer drugs.”
Study
MYC Interacts with the G9a Histone Methyltransferase to Drive Transcriptional Repression and Tumorigenesis
Authors
Tu WB, Shiah YJ, Lourenco C, Mullen PJ, Dingar D, Redel C, Tamachi A, Ba-Alawi W, Aman A, Al-Awar R, Cescon DW, Haibe-Kains B, Arrowsmith CH, Raught B, Boutros PC, Penn LZ
Funding
This work was partially supported by a grant to the Terry Fox New Frontiers Program Project Grant in Delineating therapeutic opportunities in triple-negative breast cancer